 |
|
|
|

|
LifeScienceHistory.com - Check us out on Instagram
Take Your Next Career Step with Lundback
New Drug Development Process
Steps from Test Tube to New Drug Application Review

The fully capitalized cost to develop a new biotechnology drug, including
studies conducted after receiving regulatory approval, averages $2.558 billion, according to
the Tufts Center for the Study of Drug Development (2016). When post-approval R&D costs of $312
million are included, the full, product lifecycle cost per approved drug, on average, rises to
$2.870 billion. All figures are expressed in 2013 dollars,
and the analysis was based, in part, on information provided by 10 pharmaceutical companies on 106 randomly selected drugs that
were first tested in human subjects anywhere in the world from 1995 to 2007. Notably, only
one in 1,000 compounds that enter preclinical testing make it to human testing, and only
one of the five tested in people is approved by the Food & Drug Administration (FDA).
The average time for the FDA to approve new drugs declined to 1.1 years in the 2005-07 period,
but longer average clinical phase time means combined clinical and approval time continues to
hover around eight years. The Tufts Center maintains unique
databases that provide the most detailed source of historical information available on pharmaceutical and
biopharmaceutical innovation in the United States. These databases contain data on products approved from 1963 to the present
(Tufts Center for the Study of Drug Development).
Click any of the
following boxes or words
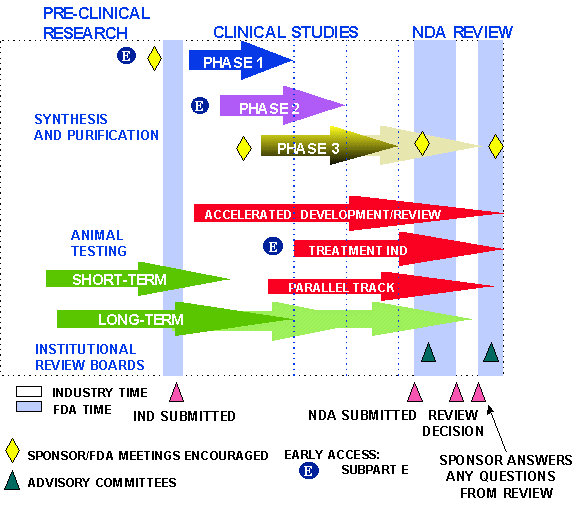
Source: U.S. Food and Drug Administration Center for Drug Evaluation and Research Handbook.
|
|
|
|
|
|